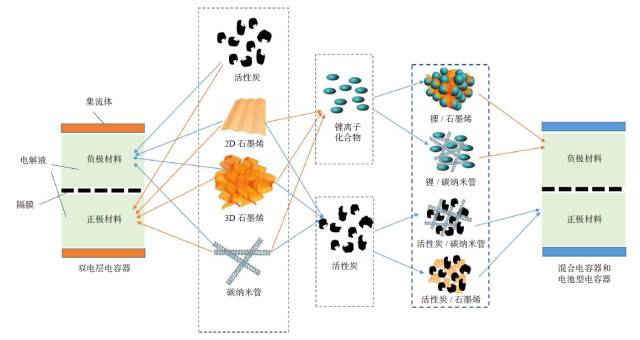
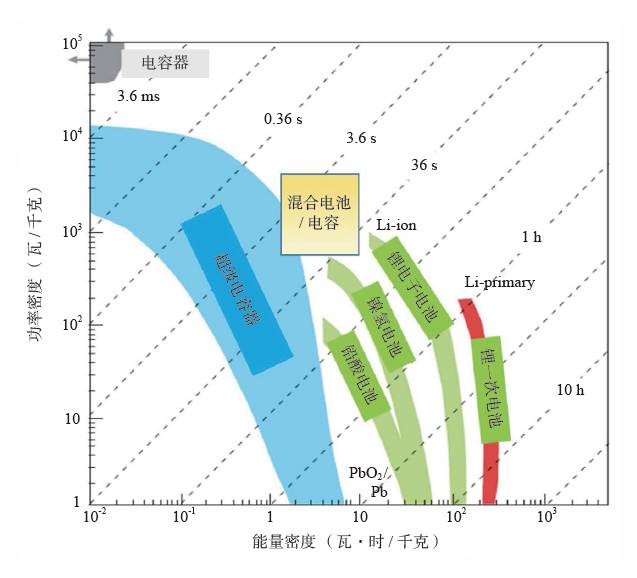
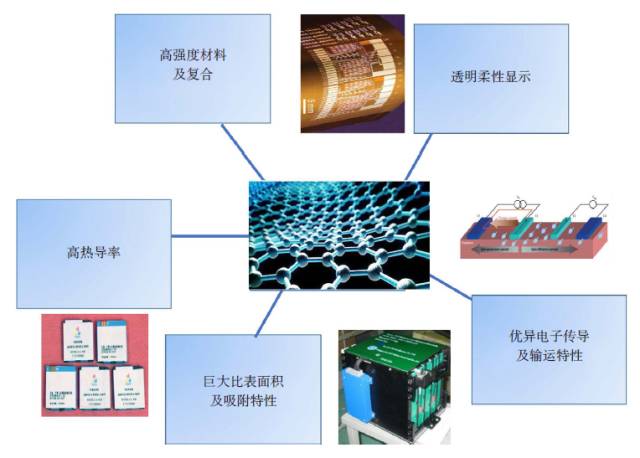
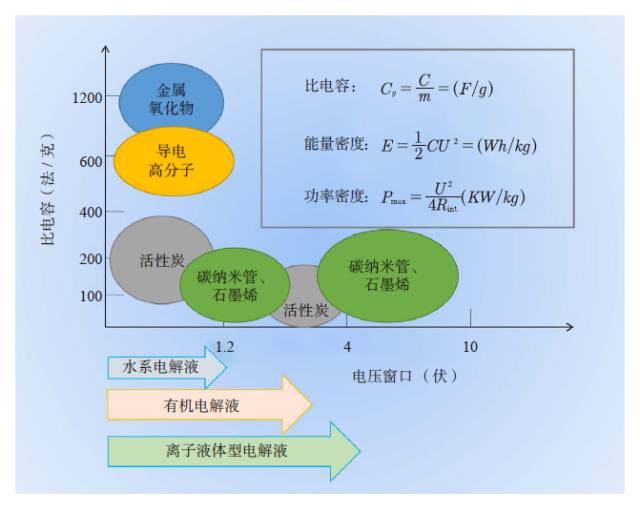
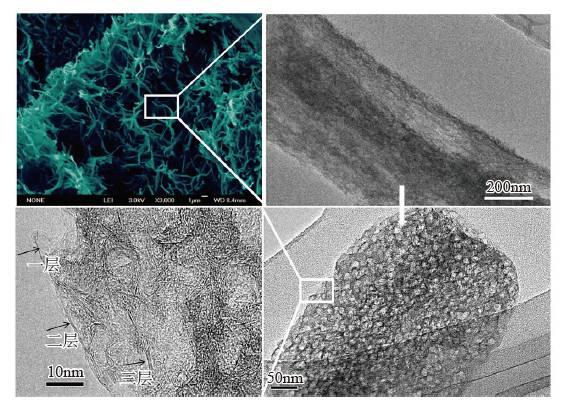
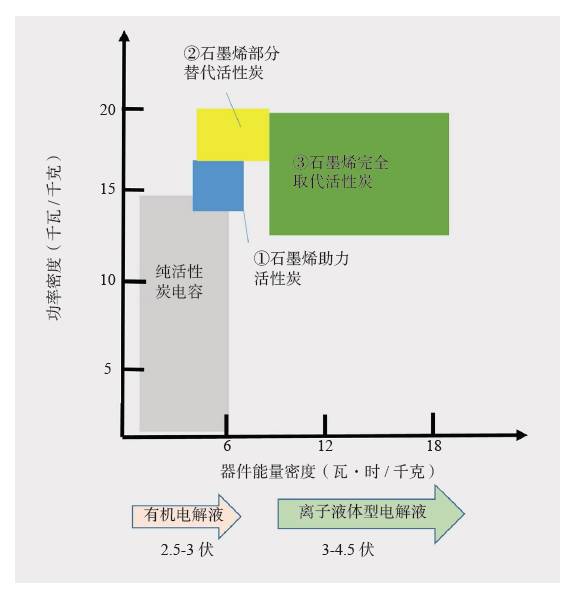
People are familiar with dry batteries and lithium-ion batteries, but they may not know much about capacitors. In fact, these energy storage devices are composed of positive and negative electrodes (anode and anode), diaphragm, current collector, electrolyte and outer casing, etc., and the electrode material is replaced, and the battery becomes a capacitor. Capacitors and supercapacitors Due to the different positive and negative materials, the performance of lithium ion batteries and capacitors is extremely different. For example, a lithium ion battery based on a lithium iron phosphate cathode material has an energy density that is more than 20 times higher than the energy density of the best supercapacitors currently on the market. The power density of a supercapacitor can be 30 to 100 times that of a lithium ion battery. If you use the analogy of runners, supercapacitors are the top 100 meters of explosive players, and lithium-ion batteries are marathon runners with outstanding endurance. The difference between capacitors and supercapacitors is mainly in the following aspects. First, the storage capacity varies depending on the type of capacitor. The smallest capacitor can only store a few microvolts and is dedicated to electronic controllers. For example, there are many capacitors in an old-fashioned radio to regulate circuit functions. A 560-ml bottle of supercapacitor can store 3,000 to 6,000 of normal power. Second, supercapacitors can provide large currents instantaneously. The initial current of heavy machinery starts is 3~6 times that of normal operation, and the general power supply system does not have such a large setting margin. The use of supercapacitors greatly simplifies the configuration of the startup system and saves costs. Therefore, the supercapacitor constitutes a module that can be used to start a blade in a wind turbine; an auxiliary crane and a start of a large truck, a light rail vehicle, and the like. In addition, supercapacitors can reversibly charge and discharge 500,000 to 1 million times, and the most advanced lithium-ion batteries are difficult to exceed 10,000 times (mostly 3,000 times), not to mention the batteries on ordinary household cars (lead-acid batteries). ) can only reversibly charge more than 300 times. Therefore, supercapacitors are often used as backup power sources for aircraft doors. Once the aircraft is in an accidental power outage, supercapacitors that are not used for a long time but are on standby can play a key role. It is also because of this extremely long service life that there are two interesting situations. (1) Although supercapacitors are far less than lithium-ion batteries in terms of storage cost per watt hour, supercapacitors can store much more electricity than lithium-ion batteries during their entire life cycle. (2) Since the supercapacitor can work cyclically for 500,000 to 1 million times, the motor vehicle equipped with the supercapacitor itself has no long life (the motor vehicle is generally scrapped for 15 years or so). Therefore, when the motor vehicle is scrapped, the supercapacitor with good performance can be removed and recycled elsewhere. The ultra-long life characteristics of supercapacitors may also explain why its current market development is far less than lithium-ion batteries. China's opportunity to develop supercapacitors Schematic diagram of capacitor and type of electrode material For a long time, due to its low energy density, supercapacitors are the supporting role of energy storage in the European and American markets. At the same time, due to the small scale of European and American cities, low population density, and saturated markets, the eyes of the world are increasingly counting on China's huge market. First, applications in energy recovery systems, such as vehicle braking, crane deceleration, etc., are traditionally mechanical energy that is wasted by friction and is completely dissipated as thermal energy. The supercapacitor, through the electromechanical conversion system, can convert the mechanical energy into electrical energy storage and release it in a pre-configured standby circuit, thereby saving energy. This market is very huge and one of the important ways to improve energy efficiency in China. At present, China has become the country with the longest international highway mileage. Many buses that travel on the highway will be the ideal tool for recycling energy from supercapacitors. At the same time, China's real estate industry is developed, and elevators in high-rise offices and residential buildings operate frequently. Using a capacitor that responds quickly is easy to start and recycle energy. Although the supercapacitor has a small amount of charge, the charging speed is fast, and it can be fully charged in half a minute to one minute. Imagine any bus station. When the passengers get on and off the vehicle, the vehicle can be fully charged and run to the next station. It can fully realize low operating energy, and is environmentally friendly and green without pollution. For the big city bus system that has been shaped in China. , has a very realistic meaning. For small and medium-sized cities with small urban areas and relatively uncongested traffic, supercapacitors with fast charging but low charging capacity will not consume their power during the crowd waiting process. Favorable choice. In comparison, charging hours require several hours of lithium-ion battery-powered electric vehicles, occupying a large number of parking lots and road resources, and the development in large cities is constrained. At the same time, supercapacitors have excellent stability and can also be used in municipal lighting systems such as street lamps, so that these lighting systems are free of repair and maintenance, which will effectively reduce the cost of electricity storage and infrastructure costs throughout the life cycle. s Choice. Automotive systems have always been a strategic application area for lithium-ion batteries and supercapacitors. At present, the point of view in Europe and America is that they are used together. That is, when an electric car starts, climbs, and brakes, a supercapacitor is used, and at a steady speed, a lithium ion battery is used. Although this energy utilization route is reasonable, it also limits the function of the supercapacitor, that is, the supercapacitor is subordinate and cannot be used as the main power source. In China, through a large number of practices, a pure supercapacitor-driven urban light rail demonstration line and a city bus bus demonstration line have been produced, effectively satisfying the instantaneous or transient rapid transport of a large number of passengers, representing a development trend. In addition, the road density of large cities in China is insufficient, the number of vehicles is large, and the absolute speed is slow. The exhaust emissions at idle speed account for the bulk of automobile emissions. Since the battery (lead-acid battery) used in cars is reliably charged and discharged too little, if a supercapacitor capable of reversible charge and discharge of 500,000 to 1 million times is used, the internal combustion engine can be extinguished at idle speed, and when needed, quickly Start up, effectively reduce exhaust emissions and achieve green transportation. Graphene boosts the development of supercapacitors General performance range diagram for different energy storage devices Small cars have a huge market. Because the space of the vehicle system is limited and the weight increase will increase the energy consumption, it is determined that the super capacitor must have the characteristics of high energy density and small volume, so increasing its energy density becomes the key to the application breakthrough. This requires upgrading existing commercial products. Taking the commercially available electric double layer capacitor as an example, most of the operating voltage is 2.7 volts, using activated carbon as the electrode material and using the organic electrolyte, the capacitance of the activated carbon electrode material is less than 200 law / gram, and the energy density of the capacitor component is less than 6~ 7 watts per hour / kilogram (or watt hour / liter). In theory, the energy density is proportional to the capacitance of the electrode material and proportional to the square of the operating voltage, which determines the increase of the operating voltage and is the key to achieving high energy density. In fact, mobile phone batteries and lithium-ion power batteries are also trying to increase the working voltage. In order to increase the working voltage, in addition to the need to replace the electrolyte with higher chemical stability, it is necessary to use a carbon electrode material with higher purity. In general, activated carbon is obtained by carbonization of coconut shell, apricot shell, petroleum coke, etc., and may contain metal impurities and impurities such as oxygen, nitrogen, phosphorus, etc. introduced during the activation process, in an aqueous electrolyte (1 volt). The redox reaction contributes to the Faraday's capacitance. However, at high voltages, these impurities can cause the electrolyte to continue to decompose, causing the device to swell and cause internal resistance to become large or even damage the device, which must be removed. At the same time, activated carbon is a microporous carbon with a "concave" structure, and the pore diameter is mostly less than 0.7 nm. For electrolytes such as organic liquids and ionic liquids, the transport of ions inside the activated carbon is like winding the labyrinth, which causes the diffusion resistance to become larger and the surface utilization rate to be lower. Graphene is a kind of SP2 hybridized carbon, and its chemical stability is much higher than that of SP3 hybridized activated carbon. At the same time, the surface of graphene is all "convex" surface, which is very beneficial to the ion proximity and adsorption or desorption of the electrolyte, achieving a rapid charge and discharge process. In particular, it can be pointed out that graphene can be prepared by high-purity hydrocarbons by chemical vapor deposition at high temperature. In principle, it can ensure a large specific surface area and ensure high purity without metal incorporation, thus providing numerous Excellent performance. In the current 13th Five-Year Plan for Renewable Energy Development, the supercapacitor urgently needs to improve its quality. Graphene materials have experienced more than ten years of development and understanding, and finally have a coincidence. A good time. Development and progress of graphene Supercapacitor cells, modules and some application areas Graphene characteristics and some application examples Graphene was first discovered in 2004 by scientists at the University of Manchester in the UK. Its appearance has caused a sensation in the international physics community, but this is not because of its well-known strength, electrical conductivity, thermal conductivity or energy storage characteristics, but because physicists did not believe in a two-dimensional planar atomic level before this. The crystal is stable. When British scientists glued a piece of good quality graphite (a macroscopic body of more than a million layers of single-layer graphene), it peeled off layer by layer and peeled off until a carbon with a thickness of only 0.12 nm was obtained. In the case of atomic single crystals, graphene exhibits a series of excellent properties such as sound, light, electricity, force, heat and magnetism, and drives the development of other atomic-scale two-dimensional materials and self-assembly techniques. Since the magic of nanotechnology in 1991, driven by the research boom of C60 and carbon nanotubes, graphene has encountered a golden age of sufficient scientific research personnel, abundant scientific funds and active wind investment. In a short span of ten years, graphene has completed a huge transformation from the "up-and-coming talent" to the "Nobel Prize darling" and achieved great results. (1) The normal direction of single-layer graphene is the material with the highest strength, and its strength is more than 100 times that of steel. Therefore, graphene can be widely used in the field of composite reinforcement of various materials. (2) Graphene, which is a non-polar material composed of a carbon-carbon six-membered ring, is macroscopically hydrophilic, and thus has a variety of surface hydrophilicity and hydrophobicity. (3) The perfect structure with flat carbon, can load a variety of metals, its performance is also well studied, can become a load research platform. (4) The single-atom layer film, which is transparent and conductive, and flexible, can be the darling of flat display and flexible devices. (5) A small hole in the regular plane of graphene can be used for forward osmosis desalination of seawater, which is a huge supplement to the current reverse osmosis seawater desalination membrane. (6) In the field of capacitors, American scientists have tried every means to set up several pieces of graphene to make microcapacitor parts, which proves that this capacitor does have ultra-fast response capability of megahertz. The premise of achieving many excellent performance and application prospects is to obtain excellent materials, so its development direction mainly includes: (1) preparing larger and larger single crystals; (2) preparing layer number and specific surface area, and increasing purity Controllable powder; (3) Direct preparation of various types of composite materials with substrates. For the graphene to be substituted for activated carbon, it belongs to the category of powder and looks like a pile of toner. For the capacitor parts to be replaced by activated carbon, it is necessary to load more and more graphene materials in a very small space, including rolling, bonding and the like. These engineering properties also require the preparation of graphene, because graphene is a two-dimensional material with a large specific surface area. Once two sheets of single-layer graphene are superposed, the huge van der Waals force will cause it to be separated again. The specific surface area is immediately reduced by 50%. The carbon nanotubes are then dispersed or grown directly between the layers of graphene sheets. Later, the template method was developed to directly grow graphene to form a honeycomb-like nanostructure. Each graphene sheet is slightly curved, naturally connected, and does not overlap. It has both a large specific surface area and a diffusion channel. Therefore, in general, the current level of graphene preparation has become closer to the demanding requirements of supercapacitor applications. Phased development of graphene-based supercapacitors Performance of electrode materials in supercapacitors and voltage window of applicable electrolyte Graphene nanofibers suitable for capacitance characteristics Due to the small output and the lack of production, the current price of high-end graphene is equivalent to that of silver, which is 4,500-6,000 yuan/kg. This objectively hinders the various applications of graphene in fields including supercapacitors. Throughout the magnifying preparation and price law of various materials, the maturity, expansion and quality of the application surface, the increase of output and the decline of price are complementary. Therefore, looking at the role of graphene in supercapacitors from a development perspective is not only in line with historical laws, but also is not speculative. The author attempts to divide the development of graphene-based capacitors into three stages. Graphene assisted activated carbon capacitor stage The characteristic of this period is that activated carbon is still the dominant electrode material of the capacitor. The amount of graphene added is usually less than 3%~4%, which only acts as a conductive agent, helping the activated carbon capacitor to reduce the internal resistance, improve the service life or appropriately increase the power density. . At present, the amount of high-end activated carbon electrode material in China is about 1000 tons/year, and the amount of graphene is about 30-40 tons/year. For example, Cnano Technology first applied carbon nanotubes to conductive agents for lithium-ion batteries in 2007. Currently, carbon nanotubes have become a preferred choice for conductive lithium-ion battery conductive agents, forming a considerable industry. . By analogy, the time period in which the graphene material realizes the function of the conductive agent does not need to be too long. Due to the small amount of graphene, based on the current slurry processing, pole piece processing and assembly process of activated carbon, the voltage platform and the test system do not need revolutionary changes, so it is the most practical and most promising breakthrough in industry. point. Part of graphene replaces activated carbon electrode material The characteristic of this period is that graphene not only acts as a conductive agent, but also functions as a part of the main electrode material, and coexists with activated carbon, and its mass fraction can fluctuate between 20% and 40%. The annual demand for graphene will increase to about 200-400 tons, which will form a relatively large industry. However, since graphene and activated carbon coexist, it will be subject to the operating voltage platform of activated carbon. In addition, due to the large volume ratio of graphene, how to maintain the surface density close to the original activated carbon on the pole piece will become the key to material processing. In order to offset the loss caused by the decrease in the density of the pole pieces, it is required to improve the structural control technology of graphene and obtain a material with a larger accessible surface area. Graphene completely replaces the stage of activated carbon electrode material If graphene completely replaces the activated carbon electrode material, it will form a market with a demand of 1,000 tons/year. The advantage is that a new electrolyte system can be used to increase the voltage of the capacitor, and the advantages of high chemical stability, high conductivity, and easy ion adsorption of graphene are exerted. However, it may cause a lower packing density of the electrode material, and increasing the density of the pole pieces will require re-architecting, which is a big challenge. In short, supercapacitor energy storage is a complex high-tech field. The requirements for electrode materials objectively exist the "barrel short board theory", that is, the water that the barrel can hold, depending on the shortest board, not the longest Board. In terms of specific surface area, purity, pore topology, electrochemical stability and electrical conductivity, graphene must win the "one" of activated carbon. Then, once the short plate of "small bulk density and large liquid absorption" of graphene is overcome, graphene can replace activated carbon. This depends on the improvement of chemical vapor deposition preparation technology and the theoretical study of the complex interaction and control between the liquid and solid soft matter levels. Graphene is used in the development of electric double layer super capacitors Electrical Products,Upvc Pipe Fitting,Pvc Pipe Fittings,Pipe Fitting Flange,Pipe Clamp Fitting FOSHAN SHUNDE LANGLI HARDWARE ELECTRICAL CO.LTD , https://www.langliplastic.com

March 09, 2023